Inquiries on the study
Mohamed Ibrahim
Associate Professor (Special Appointment) Mohamed Ibrahim
Research Institute for Synchrotron Radiation Science (HiSOR), Hiroshima University
Email: ibra2020 * hiroshima-u.ac.jp
(Note: Please replace * with @)
Inquiries on the story
Hiroshima University Public Relations Office
E-mail: koho * office.hiroshima-u.ac.jp
(Note: Please replace * with @)
Floatable beads made from chitosan and cellulose acetate and enhanced with bentonite were engineered to effectively clean oil from water. The beads showed good oil adsorption capacity while remaining easy to collect from the water surface.

(kris krüg / CC BY-SA 2.0)
Oil spills, the accidental and uncontrolled release of crude oil into the ocean, are remediated by removing oil from the environment. Typical approaches include mechanical recovery, a dangerous, labor-intensive and time-consuming process; in situ burning, which cause severe ecological disruption; bioremediation, an environmentally friendly yet slow process; chemical dispersants, which also cause severe ecological disruption; and sorbent-based approaches, which are promising but currently inadequate.
An international research team led by Hiroshima University has developed a novel sorbent for cleaning up oil spills. The sorbent, beads of chitosan/cellulose acetate@bentonite composite, has a high oil adsorption capacity and are uniquely designed to be floatable.
Their findings were published in the journal Polymers for Advanced Technologies on March 17, 2026.
Illustration of batch adsorption of medium-heavy crude by chitosan/cellulose acetate@bentonite (CS/CA@BT) composite beads (Mohamed Ibrahim / Hiroshima University)
Global oil consumption is slightly above 100 million barrels of oil a day. This consumption is supported by a large extraction and transportation infrastructure; accidents and errors lead to oil spills. Although rarer in the current day, oil spills are serious environmental threats that require immediate response.
“Many existing oil-removal materials are expensive or difficult to recover after use,” says Mohamed Ibrahim, associate professor (special appointment) at Hiroshima University’s Research Institute for Synchrotron Radiation Science (HiSOR) and researcher at the National Institute of Oceanography and Fisheries (NIOF), Egypt. “In this study, we aimed to develop a low-cost, environmentally friendly, and floatable adsorbent material based on natural polymers that can efficiently remove oil from water surfaces.”
The research team used rational design – the strategy of creating new molecules with a specific functionality – to develop the chitosan/cellulose acetate@bentonite (CS/CA@BT) composites. They fabricated the composites in the form of beads to enhance oil adsorption efficiency and environmental stability, and to possess intrinsic floatability.
The CS/CA@BT beads were fabricated to contain different amounts of bentonite: 7.69% w/w (CS/CA@BT1) and 14.29% w/w (CS/CA@BT2). Chitosan/cellulose acetate (CS/CA) beads were also fabricated as controls. Calcium carbonate (CaCO3) was used during the process to create a pore network within the beads. After fabrication, the team tested batch adsorption of medium-heavy crude oil in artificial seawater.
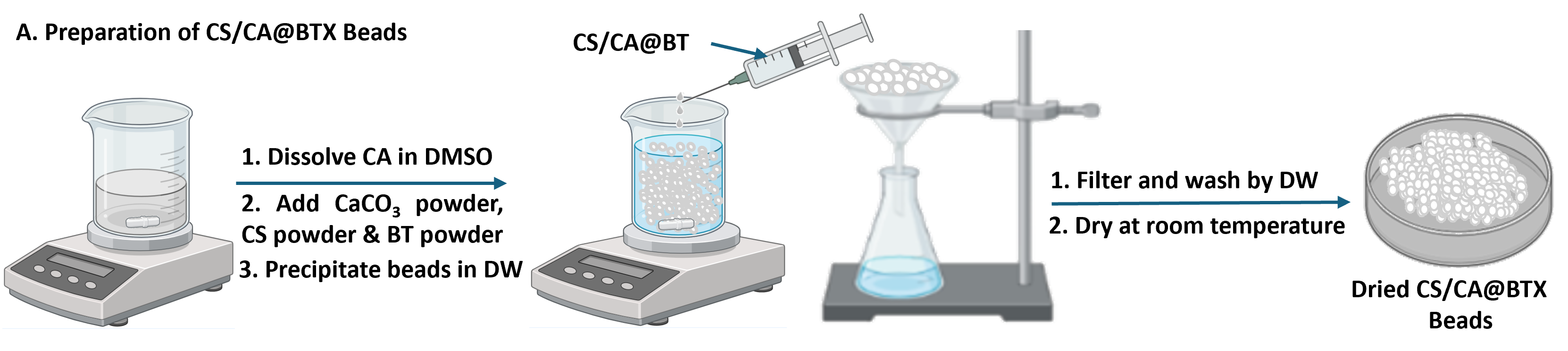
Synthesis of the chitosan/cellulose acetate@bentonite (CS/CA@BT) composite beads (Farag et al. Polymers for Advanced Technologies, March 17, 2026)
“The beads are lightweight and float on the water surface,” Ibrahim explains, “allowing them to directly capture floating oil and then be easily recovered. This makes them function like ‘tiny floating sponges’ that selectively absorb oil from water.”
Key advantages of chitosan/cellulose acetate@bentonite beads: Floatability, biodegradability, environmental compatibility, and cost-effectiveness
The composite beads were characterized using several techniques. FTIR analysis confirmed the chemical composition of the beads. Thermogravimetry analysis revealed that BT contributes to thermal resistance of the hybrid composite by acting as a thermal barrier and stabilizing agent; DSC analysis supported this by demonstrating that higher BT content enhances thermal stability of the composite. SEM showed that CaCO3 established the primary pore network in the beads, while BT - in proportion to its content - stabilizes the pore architecture, enhances surface roughness, and improves pore accessibility.
XRD analysis revealed clear differences between CS/CA@BT1 and CS/CA@BT2: the reduced BT content leads to optimal polymer-clay inter-layering in CS/CA@BT1, while the increased BT content results in an amorphous composite with partial restacking in CS/CA@BT2. Furthermore, CS/CA@BT2 was determined to have superior oleophilic properties, increasing its oil adsorption capacity. The pore structure of the beads is mesoporous (pores with diameter between 2 and 50 nm) and BT addition slightly increases the mesoporous nature of the beads.
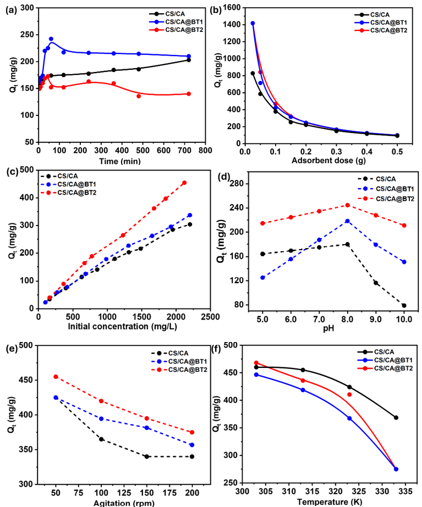
A key factor determining sorbent performance is adsorption capacity. The composite beads reached oil adsorption equilibrium within 60 minutes, significantly faster than 300 minutes for CS/CA beads. Increasing adsorbent dose (i.e., beads weight) decreased adsorption capacity, to a point. Conversely, adsorption capacity increased with oil concentration reaching a maximum of 337.3 mg/g for CS/CA@BT1 and 454.2 mg/g for CS/CA@BT2. Optimal oil adsorption occurred at a pH of 8, and agitation of 50 rpm. Finally, adsorption capacity decreased with increasing temperature, while salinity enhanced adsorption up to 10‰ for CS/CA@BT composites and 30‰ for CS/CA, after which it remained nearly constant up to 80‰. Under optimal conditions, CS/CA@BT2 exhibited the highest adsorption capacity (434.8 mg/g), demonstrating superior performance compared with unmodified CS/CA and the lower-bentonite composite.
Adsorption kinetics of the chitosan/cellulose acetate@bentonite (CS/CA@BT) composite beads under different conditions (Farag et al. Polymers for Advanced Technologies, March 17, 2026)
“Simple, low-cost biopolymer materials can be engineered to effectively clean oil from water," says Ibrahim. “Our floatable beads, made from chitosan and cellulose acetate and enhanced with bentonite, showed good oil adsorption capacity while remaining easy to collect from the water surface.”
When compared with other chitosan- and cellulose acetate-based materials for oil spill remediation, the CS/CA@BT composite beads showed superior adsorption of crude oil, particularly under environmentally relevant conditions (pH 8.0) and for realistic medium-heavy crude oils, outperforming simpler unmodified chitosan-based sorbents for crude oil, although other alternatives may exhibit higher adsorption for lighter oils. In addition, the CS/CA@BT composite beads offer key advantages such as floatability, biodegradability, environmental compatibility, and cost-effectiveness. Thus, making them promising candidates for scalable and sustainable oil spill remediation.
“The next step is to further optimize the material and evaluate its performance under real environmental conditions, including seawater and different types of oils.” Ibrahim concludes. “Our ultimate goal is to develop scalable, sustainable, and cost-effective materials that can be used in practical oil spill response systems, contributing to safer marine environments and improved environmental protection.”
Yasser F. A. Farag, Mervat A. Abdel Kawi, Hesham Zaki Ibrahim at Alexandria University, Egypt; Randa E. Khalifa at the City of Scientific Research and Technological Applications (SRTA-City), Egypt, and the Slovak Academy of Sciences, Slovakia; and, Manal G. Mahmoud and Laila A. Mohamed at the National Institute of Oceanography and Fisheries (NIOF), Egypt co-authored the study.
About the study
- Journal: Polymers for Advanced Technologies
- Title: Cost-Effective Chitosan-Cellulose Acetate Floatable Beads Embedded With Bentonite for Oil Spills Clean-Up
- Authors: Yasser F. A. Farag, Randa E. Khalifa, Manal G. Mahmoud, Mervat A. Abdel Kawi, Hesham Zaki Ibrahim, Laila A. Mohamed, Mohamed I. A. Ibrahim
- DOI: 10.1002/pat.70560
- Date: March 17, 2026

 Home
Home