E-mail: izumi-yudai*hiroshima-u.ac.jp (Please replace * with @)
Post-translational modifications of histones play a large role in helping a cell to function properly, such as repairing damaged DNA. However, it is difficult to pick apart the structural alteration of histones induced by individual modifications. By using synchrotron radiation circular dichroism spectroscopy, Yudai Izumi and his group at the Hiroshima Synchrotron Radiation Center became the first to observe what happens to the structure of H3, a type of histone protein, when it undergoes a special type of post-translational modification called K9 methylation.
In this instance, K9 dimethylation of H3 must happen so that BRCA1, a protein associated with a higher risk of breast cancer when mutated, can help repair damaged DNA. The results of this study, which were published online in the Journal of Radiation Research in December 2017, contribute toward greater understanding of how DNA damage repair works.
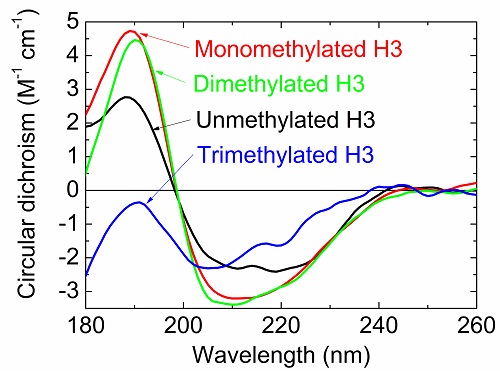
Circular dichroism spectra of unmethylated and methylated H3 proteins.
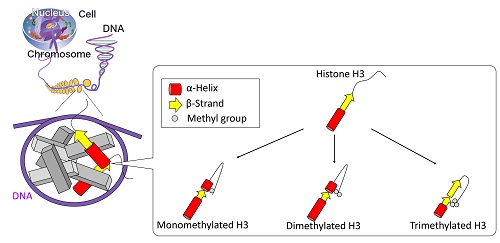
Schematic view of the proposed structures of unmethylated and methylated H3 proteins.
Reference
Title: Circular dichroism spectroscopic study on structural alterations of histones induced by post-translational modifications in DNA damage responses: Lysine-9 methylation of H3
Authors: Yudai Izumi, Koichi Matsuo, Kentaro Fujii, Akinari Yokoya, Masaki Taniguchi, and Hirofumi Namatame
Journal: Journal of Radiation Research (2018)
DOI: 10.1093/jrr/rrx068
- Profiles of Assistant Professor Yudai Izumi
- Profiles of Associate Professor Koichi Matsuo
- Profiles of Professor Hirofumi Namatame
- Profiles of Professor (Special Appointment) Masaki Taniguchi
- Official website of Hiroshima Synchrotron Radiation Center
- Find more Hiroshima University research news on our Facebook page
Assistant Professor Yudai Izumi
Hiroshima Synchrotron Radiation Center, Hiroshima University
By Margaux Phares

 Home
Home














