Using a new fluorescent mouse model with advanced imaging techniques, researchers have successfully visualized how musculoskeletal components are integrated into the functional locomotor system during embryonic development.
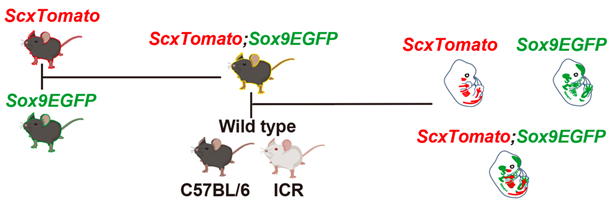
Figure 1. Breeding strategy for ScxTomato;Sox9EGFP double-reporter mice
A ScxTomato transgenic mouse was crossed with a Sox9EGFP knock-in mouse to obtain ScxTomato;Sox9EGFP reporter mice with a C57BL/6 background. The ScxTomato;Sox9EGFP reporter mouse was crossed with a wild-type C57BL/6 or ICR mouse to obtain ScxTomato, ScxTomato;Sox9EGFP, and Sox9EGFP reporter embryos. (Reproduced with permission from Development, Yu et al., 2025)
The musculoskeletal system plays an indispensable role in supporting our life, as it performs a variety of essential functions—providing structural support, enabling movement such as walking and lifting, protecting internal organs, maintaining posture, generating heat through muscle activity, and coordinating with the nervous system. One of the many complexities of this system lies in how its components—tendons, ligaments and cartilage—establish precise connections during embryonic development. Traditional methods, which often rely on histological analysis of thin tissue segments, have limitations in capturing the spatial organization and dynamic integration of these tissues in a three-dimensional (3D) context. To address this challenge, a research team from Hiroshima University has developed a novel fluorescent mouse model combined with high-resolution imaging techniques. This approach allows for clear and comprehensive visualization of how musculoskeletal components are organized and connected during organogenesis, providing new insights into the complex process of locomotor system development.
Researchers published their results in Development on March 26, 2025.
A key feature of this study is the use of a newly developed double-reporter mouse model combined with fluorescent imaging to investigate the complex processes required to form proper connections between cartilaginous and tendinous/ligamentous tissue that connect muscles and skeletal elements during embryogenesis (Figure 1). The double-reporter model enables the simultaneous tracking of two distinct reporter genes. In this study, mice expressing red and green fluorescent proteins in the expression domains of Scx and Sox9, respectively, were used to visualize the desired biological event of interest. The goal of this study is to elucidate how musculoskeletal components are integrated during organogenesis.
“Traditional methods using thin tissue sections have limitations in preserving structural integrity, which makes it difficult to study the 3D organization of these tissues. Our approach overcomes these challenges by combining tissue clearing of a newly established double fluorescent reporter mouse model with high-resolution fluorescence imaging,” said Chisa Shukunami, a professor at Hiroshima University’s Graduate School of Biomedical and Health Sciences (Figure 2).
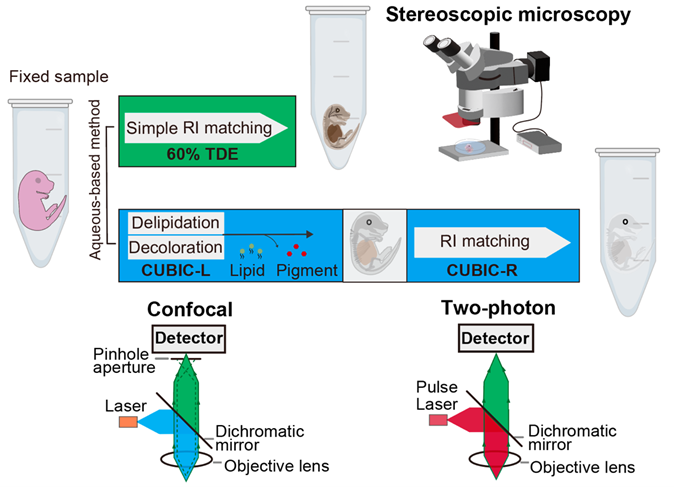
Figure 2. Tissue-clearing techniques and three-dimensional fluorescence imaging
Steps for deep-tissue 3D-imaging. Embryos or neonates fixed with 4% PFA were optically cleared with 60% TDE or tissue-clearing reagent CUBIC-L/R, and imaged using stereoscopic, confocal, or two-photon microscopy. (Reproduced with permission from Development, Yu et al., 2025)
The connection between muscle and cartilaginous primordia (early embryonic structures) via tendons, or between cartilaginous primordia via ligaments, must be precisely regulated in both spatial and temporal aspects to ensure the proper formation of a healthy and functional musculoskeletal system. To visualize this process, mouse models expressing fluorescent proteins are commonly used. Fluorescent proteins act as markers by emitting fluorescent light—causing specific cell populations to glow—and make it possible to observe where these cells are located and how they behave inside the body. This approach allows scientists to track these cells as they change position over time and to better understand how musculoskeletal tissues are assembled during development.
This study focuses on two molecules with tissue-specific expression patterns. One is Scleraxis (Scx), a basic helix-loop-helix transcription factor expressed during tendon and ligament formation. The other is SRY-box containing gene 9 (Sox9), a transcription factor essential for cartilage development. Mouse lines with ScxTomato and Sox9EGFP were crossed to obtain ScxTomato;Sox9EGFP mice (Figure 1). In the newly established double-reporter mouse model, red fluorescence highlights the Scx-expressing domains, corresponding to sites of tendon and ligament formation, while green fluorescence marks areas where Sox9 is active, indicating regions of cartilage formation (Figure 3).
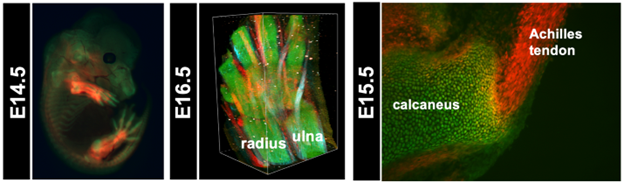
Figure 3. Visualization of cartilage and tendons/ligaments using ScxTomato;Sox9EGFP mice
In the left panel, a fluorescence stereomicroscopic image shows an optically cleared ScxTomato;Sox9EGFP mouse embryo at E14.5. The central panel presents 3D reconstructed images of the developing paw and distal forearm of a ScxTomato;Sox9EGFP at E16.5, acquired using two-photon excitation fluorescence microscopy and second harmonic generation (SHG) imaging. In this panel, tendons exhibit red fluorescence, cartilage emits green fluorescence, and mature tendons appear light blue due to SHG signals. In the right panel, a cryosection image of the Achilles tendon and calcaneus region from a ScxTomato;Sox9EGFP mouse embryo at E15.5 is shown. The Achilles tendon exhibits red fluorescence, cartilage emits green fluorescence, and yellow cells co-expressing both fluorescent proteins are localized at the attachment site. (Reproduced with permission from Development, Yu et al., 2025)
“By combining double fluorescent reporter mice with Scx-deficient mice, we demonstrated that Scx not only regulates tendon and ligament maturation, but also plays an important role in controlling muscle morphology and its attachment to cartilaginous bone primordia. These findings provide new insights into how the musculoskeletal system is established,” said Shukunami (Figures 4 and 5).
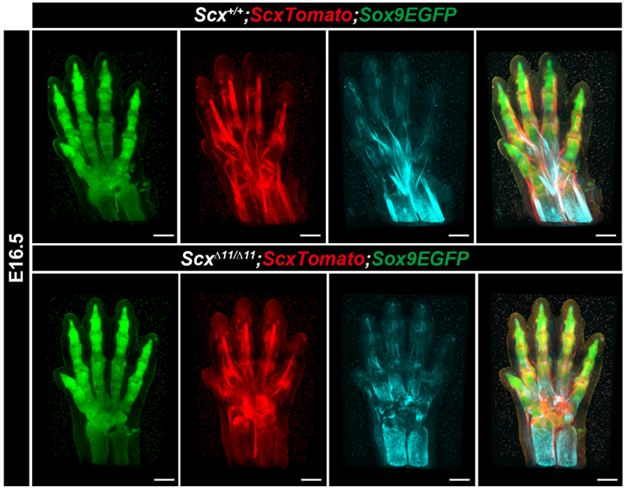
Figure 4: Fluorescent visualization of defective tendon maturation in Scx-deficient mice
Three-dimensionally reconstructed images of the developing paw and wrist of an E16.5 ScxTomato;Sox9EGFP embryo with a C57BL/6 background via two-photon excitation fluorescence (TPEF) microscopy and SHG imaging. Red and green fluorescence indicates Scx+ tendons/ligaments and Sox9+ cartilaginous bone primordia, respectively. Light blue SHG signals indicate collagen bundles in the ScxTomato;Sox9EGFP embryo. Overlay of the TPEF/SHG image. (Reproduced with permission from Development, Yu et al., 2025)
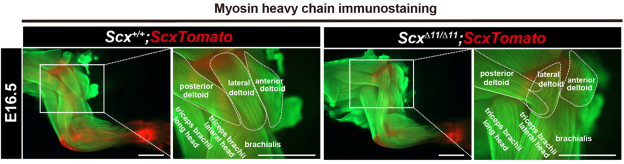
Figure 5. Misguided muscle attachment in Scx-deficient mice
Myofibers are visualized in green by anti-MHC antibody in whole-mount forelimbs of Scx+/+;ScxTomato and ScxΔ11/Δ11;ScxTomato at E16.5. Tendons are visualized in red by tdTomato expression. Magnified images of the boxed areas are shown. (Reproduced with permission from Development, Yu et al., 2025)
Additionally, it was found that the Scx+/Sox9+ cells exhibit heterogeneity in the expression levels of Scx and Sox9, meaning that there is some variability in the expression of these cells within the same cell population of cells in a given tissue (Figure 3). Such diversity in expression may be involved not only in developmental processes but also in tendon and ligament regeneration and the onset of related disorders, providing important implications for future research in musculoskeletal disorders.
Researchers hope to combine the double-fluorescent reporter mice with various genetically modified mice to investigate how the musculoskeletal system is established during embryonic development. While much of this research focuses on developing embryos, further investigation on postnatal mice—using tissue clearing techniques and 3D fluorescent imaging—may shift toward observing the postnatal maturation of tendons and ligaments. These efforts to uncover how tendons, ligaments and cartilage form connections could provide extremely valuable insights for the development of new diagnostic tools and treatment strategies for age or sport-related injuries related to these tissues.
Xinyi Yu, Shinsei Yambe, Yuki Yoshimoto, Shinnosuke Higuchi, Shigenori Miura, Kadi Hu and Chisa Shukunami of the Department of Molecular Biology and Biochemistry at Hiroshima University, Ryosuke Kawakami and Takeshi Imamura of the Department of Molecular Medicine for Pathogenesis at Ehime University, Takako Sasaki of the Department of Biochemistry II at Oita University, Hitomi Watanabe and Gen Kondoh of the Laboratory of Integrative Biological Science at Kyoto University, Haruhiko Akiyama of the Department of Orthopaedic Surgery and the Center for One Medicine Innovative at Gifu University, Ramu Sagasaki and Masafumi Inuiof the Laboratory of Animal Regeneration Systemology at Meiji University, Taiji Adachi of the Laboratory of Biomechanics at Kyoto University and Denitsa Docheva of the Department of Musculoskeletal Tissue Regeneration at University of Würzburg contributed to this research.
The Japan Society for the Promotion of Science (JSPS), the Cooperative Research Program of the Institute for Frontier Life and Medical Sciences at Kyoto University, the Ministry of Education, Culture, Sports, Science and Technology (MEXT), the Frontier Development Program for Genome Editing funded by the Doctoral Program for World-leading Innovative and Smart Education, Platforms for Advanced Technologies and Research Resources “Advanced Bioimaging Support,” and the Japan Science and Technology Agency (JST) supported this research.
About the study
Journal: Development
Title: Dynamic interactions between cartilaginous and tendinous/ligamentous primordia during musculoskeletal integration
Author: Xinyi Yu, Ryosuke Kawakami, Shinsei Yambe, Yuki Yoshimoto, Takako Sasaki, Shinnosuke Higuchi, Hitomi Watanabe, Haruhiko Akiyama, Shigenori Miura, Kadi Hu, Gen Kondoh, Ramu Sagasaki, Masafumi Inui, Taiji Adachi, Denitsa Docheva, Takeshi Imamura & Chisa Shukunami
DOI: 10.1242/dev.204512
Media Contact
Inquiries on the study
Chisa Shukunami
Professor, Graduate School of Biomedical and Health Sciences
E-mail: shukunam * hiroshima-u.ac.jp
(Note: Please replace * with @)
Inquiries on the story
Hiroshima University Public Relations Office
E-mail: koho * office.hiroshima-u.ac.jp
(Note: Please replace * with @)

 Home
Home